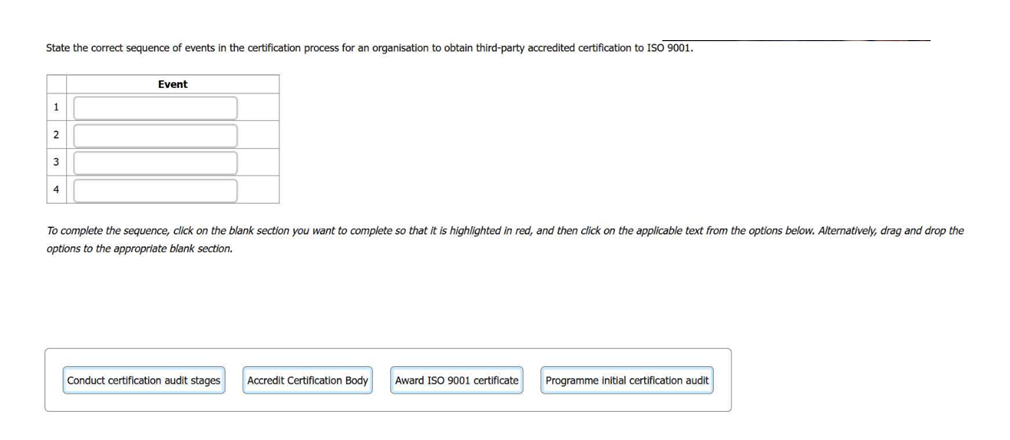
State the correct sequence of events in the certification process for an organisation to
obtain third-party accredited certification to ISO 9001.

You have been nominated audit team leader of a third-party audit. Which of the following could be the two most relevant objectives of this audit?
A. Evaluate the satisfaction interested parties
B. Evaluate the effectiveness of the management system
C. Identify the need of resources
D. Evaluate the capability of the management system to establish and achieve objectives
E. Identify opportunities for improvement
F. Evaluate the benefits obtained since the implementation of the management system
Explanation:
Evaluate the effectiveness of the management system: This objective involves verifying
that the quality management system meets the requirements of a specific standard, such
as ISO 9001:2015, and that it achieves the intended results and outcomes. The audit team
will collect and analyse audit evidence to determine the degree of conformity and
performance of the quality management system23.
•Evaluate the capability of the management system to establish and achieve objectives:
This objective involves verifying that the quality management system supports the strategic
direction and policies of the organization, and that it addresses the needs and expectations
of the interested parties. The audit team will assess the suitability, adequacy, and
alignment of the quality management system objectives, and the effectiveness of the
planning and implementation processes to achieve them23.
The other options are not the most relevant objectives of a third-party audit, according to
the web search results from my internal tool. They may be related to other aspects or types
of audits, but they are not the focus of a third-party audit.
Therefore, the correct answer is B and D.
During a third-party audit of a pharmaceutical organisation (CD9000) site of seven COVID-
19 testing laboratories in various terminals at a major international airport, you interview the CD
9000's General Manager (GM), who was accompanied by Jack, the legal compliance
expert. Jack is acting as the guide in the absence of the Technical Manager due to him
contracting COVID-19.
You: "What external and internal issues have been identified that could affect CD9000 and
its quality
management system?"
GM: "Jack guided us on this. We identified issues like probable competition of another
laboratory
organisation in the airport, legal requirements on COVID-19 continuously changing, the
shortage of
competent laboratory analysists, the epidemic declining soon, shortage of chemicals for the
analysis. It was
quite a good experience."
You: "Did you document these issues?"
GM: "No. Jack said that ISO 9001 does not require us to document these issues."
You: "How did you determine the risks associated with the issues and did you plan actions
to address
them?"
GM: "I am not sure. The Technical Manager is responsible for this process. Jack may be
able to answer this question in his absence."
Select two options for how you would respond to the General Manager's suggestion:
A. I would ask to audit the Technical Manager by phone.
B. I would ask for a different guide instead of the legal compliance expert.
C. I would look for evidence that the actions resulting from the risk assessment had been taken.
D. I would ask the consultant to leave the meeting since he is not an employee of the organisation.
E. I would delay the audit until the return of the technical manager
F. I would not accept the legal compliance expert answering the question
Explanation:
According to the ISO 9001:2015 standard, clause 4.1 requires organizations to determine
the external and internal issues that are relevant to their purpose and that affect their ability
to achieve the intended outcomes of their quality management system. Clause 6.1 requires
organizations to plan actions to address the risks and opportunities associated with these
issues. These actions must be integrated into the quality management system processes
and evaluated for effectiveness.
In this scenario, the General Manager of the pharmaceutical organization has shown a lack
of understanding and involvement in the process of identifying and addressing the external
and internal issues that affect their quality management system. The General Manager has
relied on the legal compliance expert, who is not an employee of the organization, to guide
them on this process. The General Manager has also admitted that they did not document
these issues, which is contrary to the requirement of retaining documented information on
the context of the organization. The General Manager has also delegated the responsibility
of determining and planning the actions to address the risks and opportunities to the
Technical Manager, who is absent due to COVID-19.
Based on this information, you can respond to the General Manager’s suggestion by taking
two options:
B. I would ask for a different guide instead of the legal compliance expert: You can request
to have a different guide who is an employee of the organization and who is familiar with
the quality management system processes and the external and internal issues that affect
them. The legal compliance expert may not have the necessary knowledge and authority to
answer your questions or provide you with the relevant evidence.
C. I would look for evidence that the actions resulting from the risk assessment had been
taken: You can verify whether the organization has implemented and evaluated the actions
to address the risks and opportunities associated with the external and internal issues. You
can look for evidence such as records of risk analysis, action plans, monitoring and review
results, and improvement measures.
These two options would help you to assess the conformity and effectiveness of the
organization’s quality management system with respect to the requirements of clauses 4.1
and 6.1.
XYZ Corporation is an organisation that employs 100 people. As the audit team leader, you
are conducting a certification audit at Stage 1. When reviewing the quality management system (QMS)
documentation, you find that quality objectives have been set for every employee in the
organisation except top management. The Quality Manager complains that this has created
a lot of resistance
to the QMS, and the Chief Executive is asking questions about how much it will cost. He
asks for your opinion on whether this is the correct method of setting objectives.
How would you respond with the following options? Select three.
A. Suggest asking the certification body for guidance on this matter.
B. Advise the Quality Manager to read the ISO 9001 standard and interpret in relation to the organisation's requirements.
C. Advise the Quality Manager that you will raise an opportunity for improvement if the quality objectives are not addressed properly.
D. Inform the Quality Manager that you will comment on the subject in your audit report.
E. Indicate that ISO 9001 requires a minimum of two quality objectives.
F. Suggest that all employees undertake a training course on ISO 9001.
G. Advise the Quality Manager that, as an auditor, you cannot provide advice to the organisation on how it should operate its QMS.
H. Suggest that the Quality Manager obtains external consultancy on the use of quality objectives.
Which two of the following aspects of a quality management system must the organisation continually improve?
A. Adaptability
B. Applicability
C. Effectiveness
D. Efficiency
E. Responsiveness
F. Suitability
Explanation: Effectiveness and efficiency are two of the seven quality management principles that form the basis of ISO 9001. According to ISO 9000:2015, clause 3.2.14, effectiveness is the “extent to which planned activities are realized and planned results achieved”. According to clause 3.2.15, efficiency is the “relationship between the result achieved and the resources used”. In other words, effectiveness is about doing the right things, and efficiency is about doing things right. ISO 9001:2015, clause 10.3, requires the organisation to continually improve the suitability, adequacy, and effectiveness of the quality management system. This implies that the organisation should also improve the efficiency of its processes, products, and services, as efficiency is a key factor in achieving customer satisfaction, reducing costs, and increasing profitability. Some of the ways that the organisation can improve its effectiveness and efficiency are: - Establishing SMART (specific, measurable, achievable, relevant, and time-bound) objectives and monitoring their progress - Implementing the Plan-Do-Check-Act (PDCA) cycle to plan, execute, evaluate, and improve its activities - Applying the process approach to manage its interrelated processes as a coherent system - Using evidence-based decision making to analyse data and information and take appropriate actions - Seeking feedback from customers and other interested parties and addressing their needs and expectations - Identifying and addressing risks and opportunities that can affect its performance and conformity - Encouraging innovation and creativity to find new and better ways of doing things - Benchmarking its performance against best practices and industry standards - Providing training and development opportunities for its personnel to enhance their competence and motivation 12345
Select the two statements that are true.
A. The audit team leader shall only communicate any concerns to the auditee during the closing meeting.
B. Inform the general manager if the auditor finds uncontrolled documents.
C. Where the available audit evidence indicates that the audit objectives are unattainable, the individual(s) managing the audit programme shall be immediately informed.
D. Changes to the audit scope, which become apparent during the audit, shall be approved with the auditee.
E. During the audit, the audit team leader shall periodically assess audit progress.
F. An immediate and significant risk to the audit shall be informed to the auditee and if possible to the certification body.
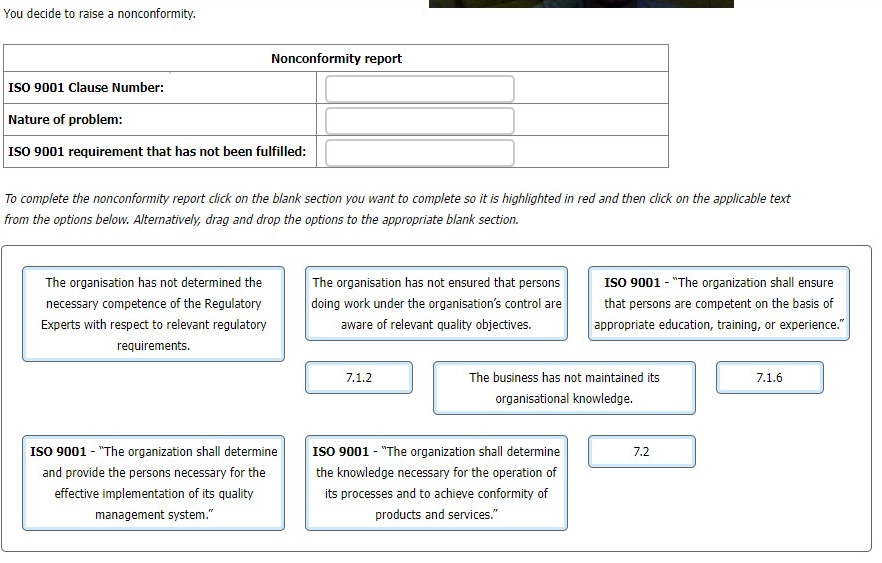
You are carrying out an audit to ISO 9001 at an organisation which offers regulatory
consultancy services to manufacturers of cosmetics.
You are interviewing the Technical Director (TD), who manages a team of regulatory
experts responsible for providing regulatory services to customers.
You: "How do you ensure your regulatory team's competence concerning regulatory
requirements is maintained?"
TD: "The two Regulatory Experts we employ full-time have years of experience of working
in the cosmetics industry."
You: "How is their regulatory competence maintained?"
TD: "They are dedicated individuals with lots of contacts in the sector."
You: "How does the business enable them to maintain their understanding of current
regulatory requirements?"
TD: "We leave that up to them."

Noitol is an organisation specialising in the design and production of e-learning training
materials for the insurance market. During an ISO 9001 audit
of the development department, the auditor asks the Head of Development about the
process used for validation of the final course design. She states that they usually ask
customers to validate the product with volunteers. She says that the feedback received
often leads to key improvements.
The auditor samples the design records for a recently completed course for the 247
Insurance organisation. Design verification was carried out but there was no validation report. The Head of Development advises that this customer required the product on an
urgent basis, so the validation stage
was omitted. When asked, the Head estimates that this occurs about 50% of the time. She
confirms that they always ask for feedback and often make changes. There is no record of
feedback in the design file for the course.
The auditor raises a nonconformity against ISO 9001. Which one of the following options is
the basis for the nonconformity?
A. 8.3.5 - The improvements made to course designs are not documented. Feedback from customers is not always actioned.
B. 8.3.2.c - Design planning does not include design validation. Design verification is part of the planning process.
C. 8.3.4.d - Design validation is not always conducted. It is omitted about half of the time.
D. 8.6 - Course materials are released without proper approval. A course for 247 Insurance was released on an urgent basis.
Explanation: Clause 8.3.4.d of ISO 9001:2015 requires that design and development
validation be performed to ensure that the resulting products or services meet the
requirements for their specified application or intended use. Validation is critical to confirm
that the product works as intended in real-world conditions.
In this case, Noitol omitted the design validation step approximately 50% of the time, which
is a direct violation of Clause 8.3.4.d. Although they collect feedback after the fact, this is
not a substitute for formal validation before the product is released. The nonconformity
arises because the process of validation was neglected, not the recording of improvements
or feedback.
Other options, such as documenting improvements (A) or issues with planning verification
(B), are important but do not directly address the primary concern: the lack of consistent
design validation before product release. Option D (8.6) concerns product release, but this
nonconformity focuses on the validation stage, not just approval for release.
During a third-party audit of a pharmaceutical organisation (CD9000) site of seven COVID-
19 testing laboratories in various terminals at
a major international airport, you interview the CD 9000's General Manager (GM), who was accompanied by Jack, the legal compliance
expert. Jack is acting as the guide in the absence of the Technical Manager due to him
contracting COVID-19.
You: "What external and internal issues have been identified that could affect CD9000 and
its quality management system?"
GM: "Jack guided us on this. We identified issues like probable competition of another
laboratory organisation in the airport, legal
requirements on COVID-19 continuously changing, the shortage of competent laboratory
analysists, the epidemic declining soon,
shortage of chemicals for the analysis. It was quite a good experience."
You: "Did you document these issues?"
GM: "No. Jack said that ISO 9001 does not require us to document these issues."
You: "How did you determine the risks associated with the issues and did you plan actions
to address them?"
GM: "I am not sure. The Technical Manager is responsible for this process. Jack may be
able to answer this question in his absence."
Select two options for how you would respond to the General Manager's suggestion:
A. I would not accept the legal compliance expert answering the question.
B. I would ask to audit the Technical Manager by phone.
C. I would delay the audit until the return of the technical manager
D. I would look for evidence that the actions resulting from the risk assessment had been taken.
E. I would ask for a different guide instead of the legal compliance expert.
F. I would ask the consultant to leave the meeting since he is not an employee of the organisation.
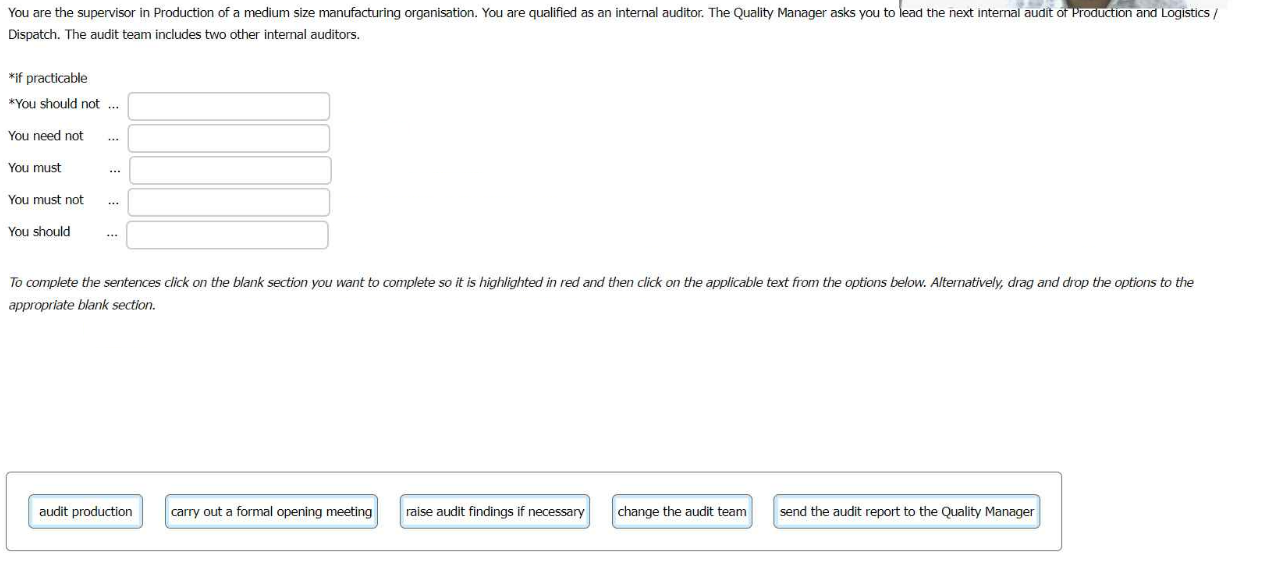
You are the supervisor in Production of a medium size manufacturing organisation. You are
qualified as an internal auditor. The Quality Manager asks you to lead the next internal
audit of Production and Logistics Dispatch. The audit team includes two other internal
auditors.
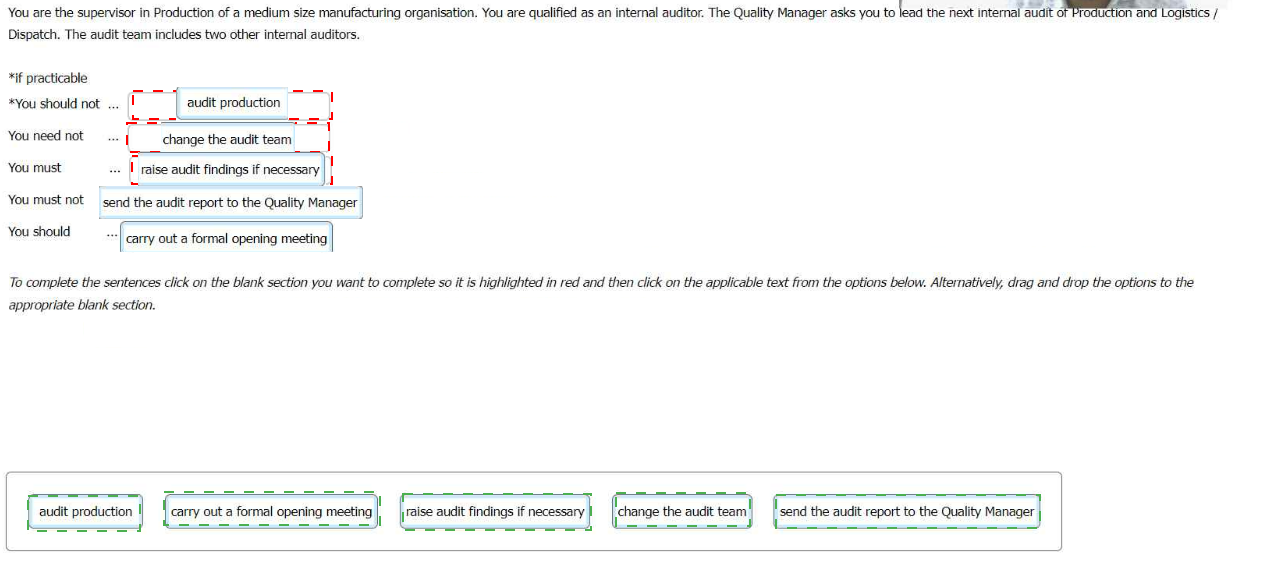
Explanation:
Here is the correct matching of actions to the statements in the context of leading
the internal audit:
If practicablecarry out a formal opening meeting
You should notaudit production (as you are a supervisor in that area, and this
would compromise audit objectivity)
You need notchange the audit team (unless there is a specific reason, such as
conflict of interest)
You mustraise audit findings if necessary (this is a key responsibility of an auditor
when nonconformities are found)
You must notsend the audit report to the Quality Manager (the audit report must be
reviewed first; it is typically part of the internal audit process to go through
necessary channels before final submission)
You shouldsend the audit report to the Quality Manager (after appropriate reviews
and approvals)
This reflects key principles of conducting an internal audit according to ISO 9001:2015,
ensuring objectivity, proper documentation, and clear reporting procedures.
TIX provides services to the informatic equipment of large organisations. They operate an
ISO 9001:2015 QMS that is being audited by an important
customer (second-party audit). During the audit, the audit team has identified two
nonconformities. When preparing the Closing meeting, the audit
team discussed and agreed both nonconformities with TIX's quality manager. The Closing
meeting was planned for 6pm with the general manager,
quality manager and service manager at the meeting room.
At 6pm, when the audit team enters the meeting room, only two people are present and
waiting for them: the Health and Safety supervisor and the
warehouse supervisor. Neither have participated in the audit.
The dialogue among them is as follows:
Audit team leader: "Good evening, could you please inform the three managers that we are
ready to start with the Closing meeting?"
Health and Safety supervisor: "Good evening. We are sorry to inform you that the general
manager was involved in a serious car accident, and
the other two managers have had to leave urgently to attend the emergency."
Warehouse supervisor: "They have asked us to listen to what you need to say and to sign
whatever we need to sign. We also have a message
from them about the two nonconformities. They wanted us to ask you if you could contact them in a couple of days to determine how to proceed."
Which one of the following options would be your preferred response to the final comment
made by the warehouse manager?
A. Sorry, but we cannot proceed with the Closing meeting. So, we are leaving now, and please tell the quality manager that I will phone him tomorrow early in the morning.
B. We will hear what you were asked to tell us and will ask you to sign the nonconformity reports as evidence that you have accepted them. Please ask the managers to contact us as soon as the emergency is over to agree on a new date to complete the Closing meeting.
C. We will hear what you were asked to tell us and will leave copies of the nonconformity reports that have been agreed with the quality manager. Please tell the managers that we will consider this as the Closing meeting and that the individual(s) managing the audit programme will send the full report in five days.
D. We will hear what you were asked to tell us and will then leave. Please ask the managers to contact us as soon as the emergency is over to agree on a new date to carry out the Closing meeting.
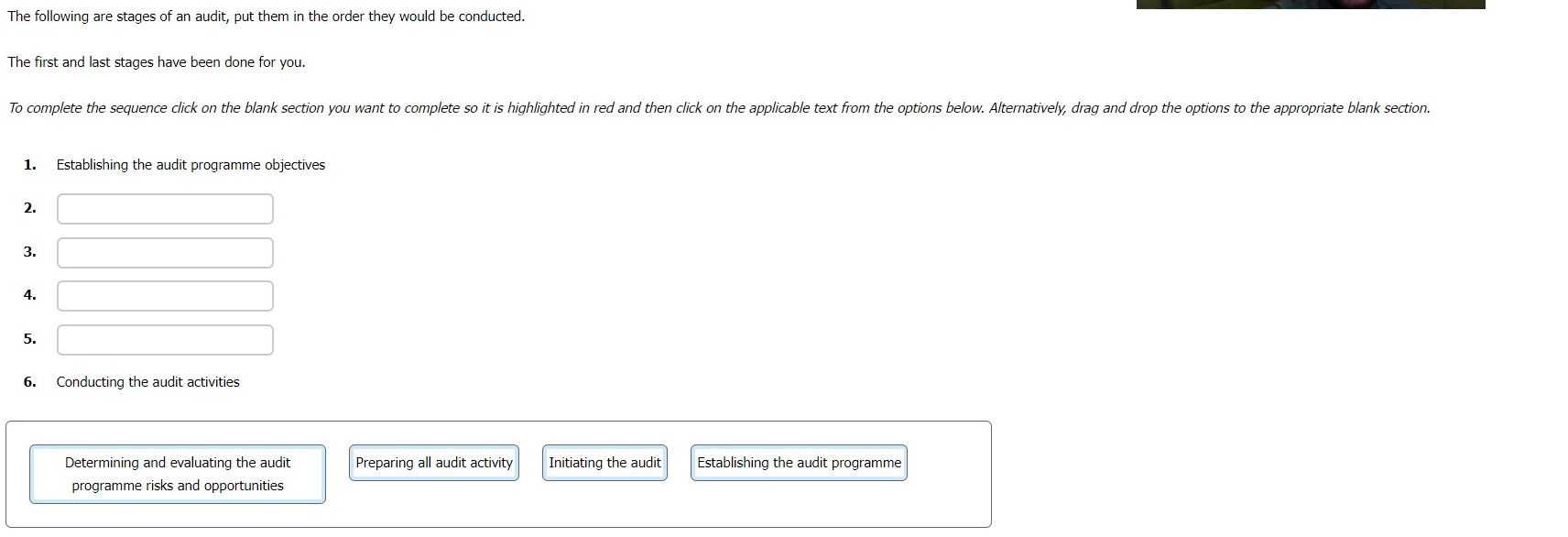
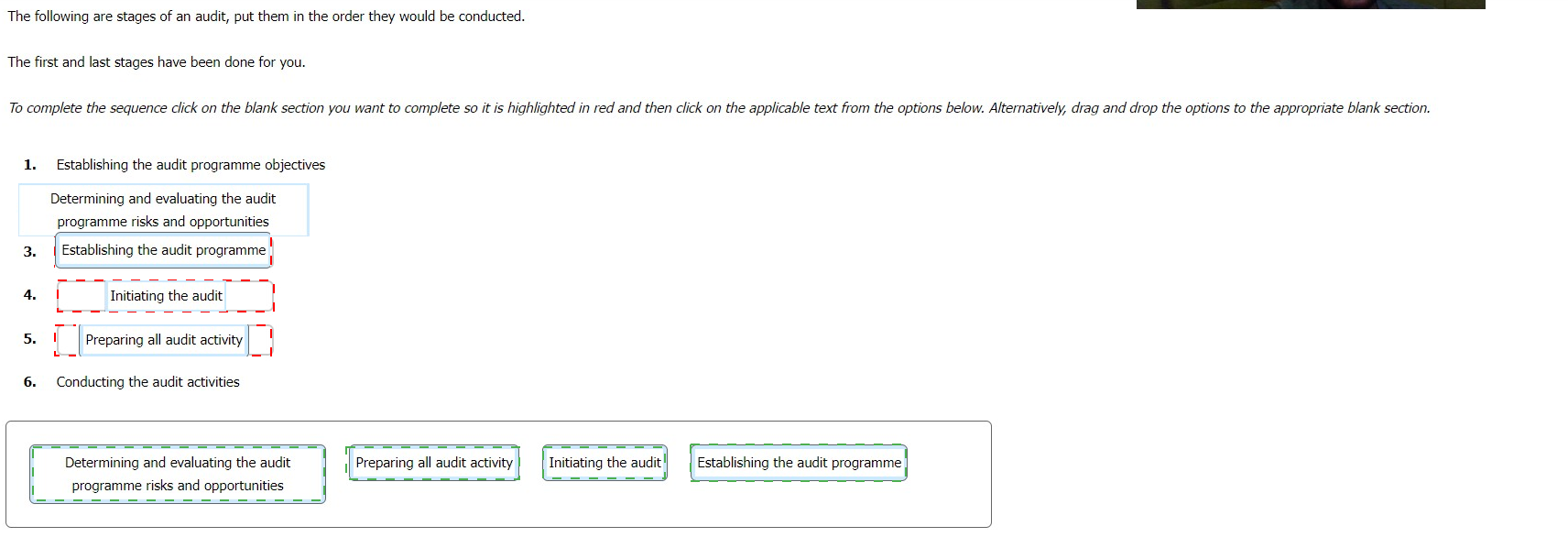
The following are stages of an audit, put them in the order they would be conducted.
| Page 3 out of 18 Pages |
| 123456 |
| ISO-9001-Lead-Auditor Practice Test Home |
Real-World Scenario Mastery: Our ISO-9001-Lead-Auditor practice exam don't just test definitions. They present you with the same complex, scenario-based problems you'll encounter on the actual exam.
Strategic Weakness Identification: Each practice session reveals exactly where you stand. Discover which domains need more attention, before QMS ISO 9001:2015 Lead Auditor Exam exam day arrives.
Confidence Through Familiarity: There's no substitute for knowing what to expect. When you've worked through our comprehensive ISO-9001-Lead-Auditor practice exam questions pool covering all topics, the real exam feels like just another practice session.